Modern, User-Friendly eConsent for Clinical Trials
Deliver fully informed consent with a digital experience that's intuitive for participants, easy to set up for study teams, and built to support compliance and retention from the start.
Request a Demo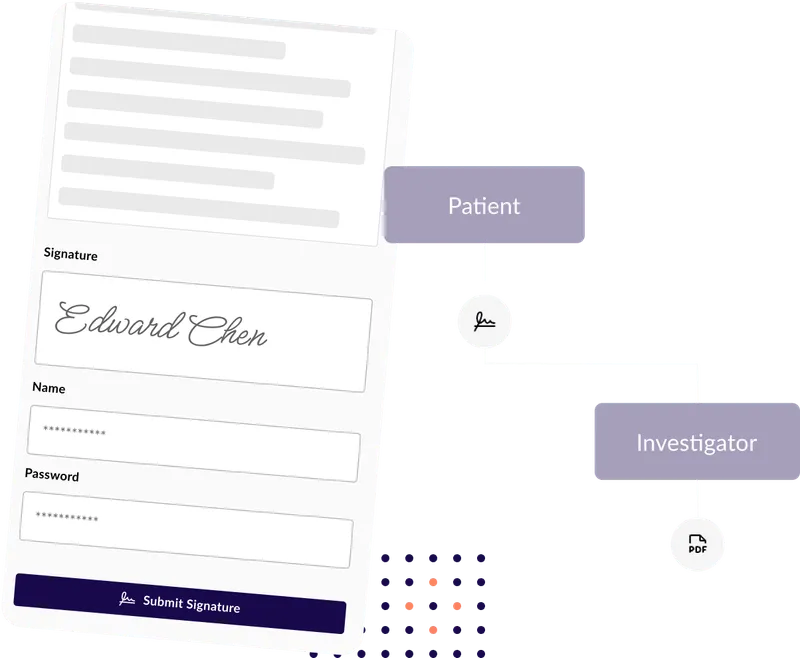
Streamlined eConsent for Understanding and Compliance
Simple for Participants
Streamlined, multimedia consent experience across any device
Digital Signature Capture
Secure, compliant electronic signatures with verification and audit trails
Multi-language by Design
Support for 40+ languages and localized formatting
Interactive Consent Forms
Video, images, and quizzes to help participants understand before signing
Integrated Experience
Seamlessly connects with scheduling, notifications, ePRO, and more
Built for Compliance
Meets 21 CFR Part 11 and GDPR standards with full audit trails
How It Works
For Participants
Step-by-step consent with video, images, and plain language—on any device
For Study Teams
Drag-and-drop builder with version control, localization, and re-consent support
Fully Integrated
Consent connects with recruitment, engagement, and ePRO in one structured platform
