Top Clinical Research Software in 2026: 20 Best Tools

Clinical research software is a category of digital tools designed to manage, execute, and analyze the vast amounts of data generated during clinical trials. From initial study design to final regulatory submission, these platforms provide the digital infrastructure for modern medical discovery. They replace cumbersome paper based methods with streamlined, compliant, and efficient workflows, empowering research teams to focus on science, not administration. This technology is fundamental to bringing new therapies, diagnostics, and medical devices to patients faster and more safely.
Why Clinical Research Software Matters
The complexity and cost of clinical trials are staggering. A single Phase 3 trial can involve thousands of patients across dozens of sites and generate millions of data points. Using the right clinical research software is not just a matter of convenience, it is a strategic necessity for overcoming a number of industry wide challenges.
- Accelerating Timelines: Delays are incredibly costly. The average time to bring a new drug to market can be over a decade. Efficient software automates manual tasks, streamlines communication, and provides real time oversight, helping to shorten these timelines.
- Improving Data Quality: Manual data entry is prone to errors, which can compromise the integrity of a study. Clinical research software enforces data standards through features like electronic data capture (EDC), ensuring information is accurate, complete, and compliant from the start.
- Enhancing Participant Experience: High dropout rates can jeopardize a trial. Modern software improves the participant journey with tools for easy electronic consent (eConsent), patient reported outcomes (ePRO), telehealth visits, and automated payment, which boosts retention.
- Expanding Access and Diversity: Traditional trials often struggle to recruit diverse populations due to geographic limitations. Platforms that support decentralized clinical trials (DCT) and hybrid models, like the Curebase Omnisite model, allow patients to participate from anywhere, dramatically widening the recruitment pool.
Core Software Components and Systems
A comprehensive clinical research software platform is not a single tool but an integrated suite of modules. While some vendors offer standalone solutions, the trend is toward unified platforms that provide a single source of truth for the entire study.
A Clinical Data Management System (CDMS) serves as the secure, validated repository for all data collected during a trial. While historically a separate category, today most leading platforms integrate CDMS capabilities directly into their core systems, creating a single source of truth for study data.
Key Software Modules
- Electronic Data Capture (EDC): The digital backbone for collecting patient data in a structured, compliant format. It is the primary component of a modern CDMS.
- Electronic Patient Reported Outcomes (ePRO) and eCOA: Allows patients to report their symptoms and experiences directly through a mobile app or web portal. AI powered ePRO tools can further enhance data collection and patient engagement.
- Electronic Consent (eConsent): Digitizes the informed consent process with multimedia explanations and secure electronic signatures, ensuring full participant understanding and documentation.
- Clinical Trial Management System (CTMS): The operational hub for managing study milestones, site performance, budgets, and regulatory documents.
Platforms like Curebase’s eClinical software combine these core software modules with execution services, offering sponsors a more cohesive way to run decentralized or hybrid studies without stitching together multiple vendors.
Implementation and Selection Criteria
Choosing the right clinical research software requires a careful evaluation of your study’s specific needs, your team’s technical capabilities, and the vendor’s track record.
What to Look For
- Regulatory Compliance: The software must be fully compliant with regulations like 21 CFR Part 11 and GDPR. Ask vendors for their validation documentation and recent audit outcomes.
- Integration Capabilities: The platform should seamlessly integrate with other systems, such as electronic health records (EHR) and laboratory information systems, to create a unified data flow.
- Scalability and Flexibility: Can the software support a small Phase 1 study as well as a large, global Phase 3 trial? Look for a flexible model that can adapt to different study designs, including traditional, hybrid, and fully decentralized trials.
- User Experience: The software must be intuitive for all users, including sponsors, site staff, and patients. A poor user experience for participants can lead to low adherence and high dropout rates.
- Support and Maintenance: A vendor’s commitment does not end after implementation. Evaluate their support model, availability, and service level agreements (SLAs). Reliable maintenance and responsive technical support are crucial for minimizing downtime and resolving issues quickly, ensuring your study stays on track.
How to Select the Right EDC and CDMS Solution
Since the EDC system is the core of clinical data collection, its selection is critical. Modern EDC platforms have largely absorbed the functions of a traditional Clinical Data Management System (CDMS), so the selection criteria for both are deeply intertwined. A thoughtful evaluation ensures data integrity, operational efficiency, and regulatory compliance.
EDC and CDMS Evaluation Criteria
When evaluating an EDC or an integrated platform with CDMS capabilities, focus on these key functional areas:
- Study Build and Design: How quickly and easily can you build and deploy a study? Look for intuitive form builders, reusable global libraries, and the ability to make mid study amendments without downtime.
- Data Entry and Cleaning: The system should facilitate clean data from the start. Key features include a user friendly interface for site staff, robust edit checks, offline data capture capabilities, and an efficient query management workflow.
- System Integration: Assess the platform’s ability to connect with other eClinical systems. Look for a robust API, support for CDISC standards (like ODM and CDASH), and proven integrations with EHR, CTMS, and lab systems.
- Reporting and Analytics: Real time data visibility is essential. The software should offer standard reports, customizable dashboards for data review and risk based monitoring, and flexible data export options in various formats (e.g., SAS, CSV).
- Participant Facing Features: For modern trials, especially DCTs, the EDC must connect seamlessly with participant tools. Evaluate the integrated ePRO, eConsent, and scheduling modules for usability and reliability.
A Framework for Comparing EDC Vendors
Moving from a long list of features to a final decision requires a structured vendor comparison. Instead of a simple checklist, use a framework that assesses each vendor holistically.
- Technical and Functional Fit: Create a scorecard based on the evaluation criteria above. Rate each vendor on how well their platform meets your specific protocol and workflow requirements. Does their system excel in the areas most critical to your study’s success?
- Vendor Experience and Reliability: Go beyond the demo. Ask for case studies or references from organizations that have run similar trials. Investigate their history in your specific therapeutic area and their track record with regulatory inspections.
- Support and Partnership Model: Evaluate the vendor’s implementation process, training programs, and ongoing support. Is their support team responsive and knowledgeable? Do they act as a true partner invested in your study’s success or simply a software provider?
- Total Cost of Ownership: Look beyond the initial license fee. Factor in all potential costs, including setup, validation, training, support, and any charges for mid study changes or data exports. A seemingly cheaper option may become more expensive over the lifecycle of the trial.
Beyond CTMS: Creating a Unified Work Management Ecosystem
While a CTMS is vital for operational oversight, modern clinical research demands a more connected ecosystem. The most advanced clinical research software acts as a central platform that integrates data and workflows from various sources. This includes everything from wearable devices and telehealth platforms to mobile phlebotomy services for at home biospecimen collection.
This approach extends to integrating with general project management tools. By connecting a CTMS to platforms like Asana or Jira, teams can sync clinical milestones with broader project timelines and manage resources more effectively. This creates a holistic view where clinical operations and corporate project management are aligned. This level of integration provides sponsors and CROs with real time visibility and reporting into every aspect of the study, enabling proactive decision making and risk management.
Top 20 Clinical Research Software Tools for 2026
As the clinical research landscape continues to evolve, the right software is essential for streamlining operations, ensuring data quality, and accelerating timelines. To help you navigate the options, we’ve curated a list of the top 20 clinical research software tools poised to make a significant impact in 2026. This selection covers the entire clinical trial lifecycle, from initial study design to regulatory submission.
1. Curebase, Inc, an AI native eClinical platform for decentralized trials

Curebase brings an AI native eClinical stack to sponsors and CROs, unifying EDC, ePRO/eCOA, and multimedia eConsent with telemedicine, participant payments, and portals for sites and sponsors. Built for 21 CFR Part 11/GxP compliance, it offers API/SSO integrations and connects to digital endpoints and specialty devices.
- Why it stands out
- AI assisted study designer speeds form builds, translations, and QC.
- Unified EDC + ePRO/eCOA + eConsent keeps oversight in one workspace.
- BYOD ePRO with offline capture and smart reminders drives compliance.
- Integrated scheduling and telehealth streamline remote visit execution.
- Single mobile app centralizes tasks, messages, and compensation.
- Ideal trial fit
Designed for decentralized and hybrid studies where participant usability wins, particularly digital therapeutics, device, diagnostics, and mental health programs seeking high engagement and clean, real time data. - Pricing snapshot
Quote based; priced per study and configured modules.
DCT and unified eClinical platforms
The industry’s shift towards decentralized clinical trials (DCT) has spurred the demand for comprehensive, all in one solutions that can manage various trial activities from a single interface. These unified eClinical platforms are designed to support modern trial models by integrating everything from data capture and patient engagement to site management. The tools in this category represent the forefront of integrated technology, offering robust capabilities to run complex, patient centric studies.
1. Zelta, an enterprise eClinical platform with EDC, eCOA, and RTSM
Zelta (formerly IBM Clinical Development) offers a cloud eClinical platform for sponsors, CROs, and sites that brings EDC, eCOA/ePRO, eConsent, and RTSM onto a single code base. The GxP validated, 21 CFR Part 11 system integrates via APIs, SSO, and CDISC outputs for enterprise programs.
- Why it stands out
- Unified data flow across EDC, eCOA, and eConsent.
- RTSM with predictive resupply and dynamic randomization.
- Fast deployment of mid study amendments to limit downtime.
- AI assisted MedDRA/WHO Drug coding to speed data cleaning.
- CDISC/ODM outputs and robust partner integrations.
- Ideal trial fit
Built for biopharma, device, and diagnostics teams running Phase I to IV and global DCT/hybrid trials that need scale, speed, and governance. - Pricing snapshot
Quote based via enterprise subscription or pay per use.
2. encapsia, a unified EDC and offline eSource platform with live analytics

Encapsia delivers a cloud clinical data suite for sponsors, CROs, and sites that blends web EDC, offline iPad eSource, and live RBQM/analytics. Designed for DCT/hybrid models, it meets 21 CFR Part 11 and integrates with RTSM/IRT and external systems via APIs.
- Why it stands out
- Zero downtime amendments across unified EDC and eSource.
- Home visit mode and mobile image capture for decentralized workflows.
- Embedded RTSM/IRT integration for dispensing and randomization.
- Live RBQM dashboards with dedicated lab/data review apps.
- Ideal trial fit
Biotech and pharma running complex global studies, such as oncology and rare disease, seeking real time oversight and smooth field/home visit execution. - Pricing snapshot
Quote based, available per study or per program.
3. Clario, endpoint technology for eCOA, imaging, and cardiac safety
Clario focuses on high fidelity endpoint collection for global sponsors and CROs, spanning eCOA, imaging, cardiac, and respiratory data. The 21 CFR Part 11 platform supports BYOD and clinic devices, integrates via device APIs, and uses federated SSO for secure access at scale.
- Why it stands out
- eCOA across BYOD, web, and telehealth to capture outcomes anywhere.
- Central imaging platform with cloud viewer for near real time reads.
- Cardiac safety and respiratory endpoints (ECG/spirometry) in one place.
- Digital mobility outcomes via wearables and precision motion sensors.
- Federated SSO and policy driven access controls.
- Ideal trial fit
Endpoint heavy Phase II to III decentralized or hybrid trials needing centralized reads and reliable at home capture for global populations. - Pricing snapshot
Quote based, tailored to the study’s endpoint mix.
Electronic Data Capture (EDC) systems
At the heart of any clinical trial is the collection of clean, secure, and reliable data, which is the primary role of an Electronic Data Capture (EDC) system. While many unified platforms include EDC functionality, a number of leading standalone systems offer powerful, specialized features for this critical task. The following EDC systems are recognized for their user friendliness, robust data validation capabilities, and seamless integration with other eClinical tools.
1. OpenClinica, an EDC/ePRO platform with EHR to EDC eSource
OpenClinica is a cloud eClinical platform used by sponsors, CROs, and sites to run compliant trials end to end. It combines EDC, ePRO/eCOA, and eConsent with EHR to EDC eSource. The system is 21 CFR Part 11/GxP validated and integrates via REST APIs, SSO, CDISC ODM, and SMART on FHIR.
- Why it stands out
- Drag and drop EDC builder with versioning and integrated randomization.
- BYOD ePRO/eCOA with multilingual support and automated reminders.
- Multimedia eConsent, comprehension checks, and compliant e signatures.
- SMART on FHIR connector to auto populate eCRFs from the EHR.
- Oversight dashboards and analytics for RBQM style monitoring.
- Ideal trial fit
A strong fit for sponsors/CROs across Phase I to IV and decentralized/hybrid designs. Particularly valuable for teams prioritizing rapid builds and reduced SDV via automated EHR eSource. - Pricing snapshot
Quote based per study/module; free trial available.
2. Medrio, a rapid build EDC with eConsent, ePRO, and RTSM
Medrio delivers a validated eClinical suite for sponsors, CROs, and sites, unifying EDC, ePRO/eCOA, eConsent, and RTSM. It emphasizes no code, zero downtime builds, adheres to 21 CFR Part 11/GxP, and integrates through APIs/SSO with CDISC aligned outputs for smooth downstream analysis.
- Why it stands out
- No code EDC with fast study start up and mid study flexibility.
- AI enabled reporting and dashboards for faster signal detection.
- Multimedia eConsent with compliant audit trails for hybrid workflows.
- Integrated ePRO/eCOA and RTSM to align patient and supply data.
- Ideal trial fit
Biopharma and medtech teams running Phase I to IV drug, device, or DTx programs needing rapid stand up, agile amendments, and unified patient facing modules. - Pricing snapshot
Quote based; contact Medrio for a tailored proposal.
3. Castor EDC, a unified EDC with native eConsent and ePRO
Castor centralizes data capture for sponsors and CROs with cloud EDC, integrated eConsent, and ePRO/eCOA. Fully 21 CFR Part 11/GxP compliant, it supports REST APIs, enterprise SSO, and CDISC/FHIR for traditional, hybrid, and decentralized studies.
- Why it stands out
- FormSync version control enables safe mid study CRF updates.
- Global BYOD ePRO/eCOA with multi channel reminders boosts adherence.
- Multimedia eConsent with video, comprehension, and audit trail.
- Centralized monitoring with configurable edit checks and SDV.
- Open REST API, SSO, and CDISC/FHIR interoperability.
- Ideal trial fit
Biopharma and medtech teams running hybrid/DCT trials that demand fast builds, global reach, and strong participant compliance via BYOD. - Pricing snapshot
Modular, quote based pricing; free trial offered.
4. ClinCapture, a private cloud EDC with ePRO, eConsent, and RTSM
ClinCapture provides a validated, private cloud EDC suite for sponsors and CROs delivering decentralized trials. With integrated ePRO, eConsent, and RTSM, it maintains 21 CFR Part 11 compliance and connects through APIs/SSO with CDISC exports for external analysis and reporting.
- Why it stands out
- Flexible, code optional study builds and amendment support.
- Unified ePRO/eCOA and eConsent for remote patient workflows.
- Built in RTSM enables secure, blinded management.
- RBM dashboards and eTMF streamline oversight and audit readiness.
- REST APIs and CDISC exports for interoperability.
- Ideal trial fit
Sponsors/CROs in device or biotech running decentralized studies requiring rapid configuration, multilingual reach, and efficient central monitoring. - Pricing snapshot
Quote based; add ons priced by module.
5. Dacima Clinical Suite, an all in one EDC with IWRS and ePRO
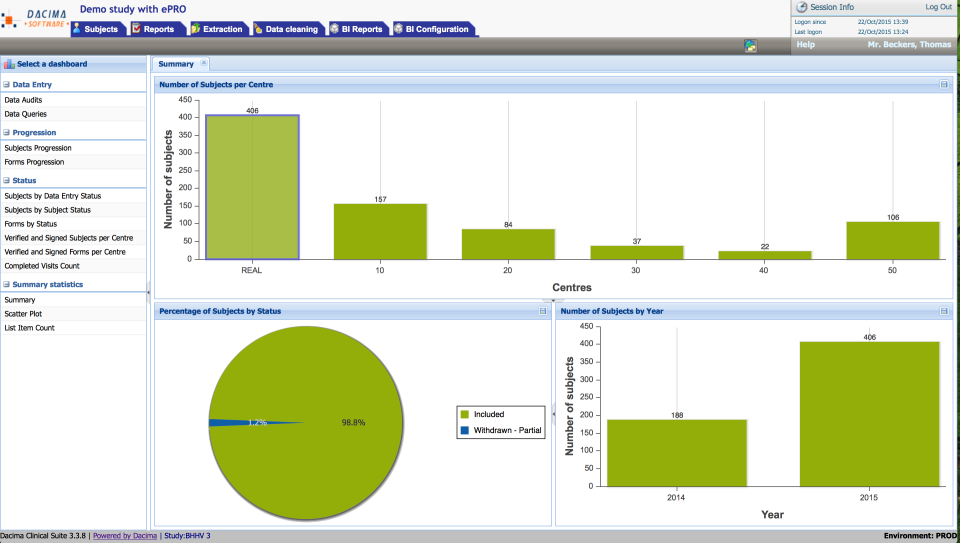
Dacima Clinical Suite serves sponsors, CROs, and academic teams with EDC, ePRO/eCOA, eConsent, and IWRS for supply management. The platform is 21 CFR Part 11 compliant, deploys SaaS or on prem, and integrates via CDISC ready APIs to downstream systems.
- Why it stands out
- Configurable EDC with offline capture and complete audit trails.
- ePRO/eCOA with automated scheduling and reminders to reduce missing data.
- Multimedia eConsent for in person or remote consenting.
- Built in IWRS connects randomization to drug supply.
- Ideal trial fit
Small to mid market sponsors, CROs, and academic researchers running Phase II to IV, device, or decentralized studies that need integrated IWRS without stitching multiple vendors. - Pricing snapshot
Quote based with per study, academic, and enterprise options.
6. MainEDC, an EDC platform with built in IWRS/RTSM and eConsent
MainEDC offers a private cloud eClinical suite for sponsors, CROs, and sites, combining EDC, IWRS/RTSM, ePRO/eCOA, and eConsent. The validated, GxP/21 CFR Part 11 environment integrates with CTMS via APIs and supports CDISC standards for clean data exchange.
- Why it stands out
- Drag and drop eCRF designer with native lab handling.
- IWRS/RTSM for randomization, dosing, and supply management.
- ePRO/eCOA with visit scheduling and SMS/push reminders.
- Risk based monitoring dashboards for data quality oversight.
- Blockchain backed audit trail in a validated private cloud.
- Ideal trial fit
Sponsors/CROs across Phase I to IV drug/device programs, including DCT/hybrid, where unified RTSM and rapid deployment reduce vendor sprawl. - Pricing snapshot
Quote based per study; free trial available.
7. PRELUDE, a no code EDC with offline capture and RTSM
Prelude delivers a cloud EDC platform for sponsors, CROs, and academic teams, bundling a no code builder, ePRO, eConsent, and native RTSM. Fully validated and 21 CFR Part 11 compliant, it integrates via APIs and supports offline capture for field or remote sites.
- Why it stands out
- No code design accelerates build, validation, and versioning.
- Seamless mid study amendments without downtime or data loss.
- Native ePRO app with automated reminders for longitudinal data.
- Offline capture with automatic sync for low connectivity settings.
- Integrated RTSM for randomization and supply logistics.
- Ideal trial fit
Biotechs, medtech, and academic teams executing complex hybrid or field based studies that require rapid configuration and offline resiliency. - Pricing snapshot
Quote based and tailored to study scope.
CTMS and study operations management
Effectively managing the logistical complexities of a clinical trial, from site selection and budget tracking to milestone reporting, is crucial for operational success. Clinical Trial Management Systems (CTMS) serve as the central command center for overseeing these administrative and project management functions, providing robust tools for project tracking and resource allocation. The software in this section provides the essential tools for ensuring studies stay on schedule and within budget while maintaining clear oversight across all sites.
1. RealTime CTMS, a site centric CTMS with eSource and eReg
RealTime CTMS centralizes day to day site operations for research sites, networks, and sponsors/CROs. The cloud suite includes CTMS, eSource, eReg/eISF, eConsent, and payments within a 21 CFR Part 11 framework, plus SSO, APIs, and EDC Connect to cut duplicate entry.
- Why it stands out
- Automated visit calendars and text reminders to reduce no shows.
- Built in recruitment tools with study pages and prescreen forms.
- Full financials: budgets, invoicing, receivables, and reporting.
- Unified eSource, eReg/eISF, and remote eConsent for inspection ready records.
- EDC Connect to streamline eSource to EDC data flow.
- Ideal trial fit
Independent sites and networks managing Phase II to IV portfolios, especially hybrid programs needing remote consenting, participant engagement, and clean source data. - Pricing snapshot
Quote based per module and operational scope.
2. Clinical Conductor, a CTMS for site networks and health systems
Clinical Conductor is a site focused CTMS that unifies study operations, recruitment pipelines, and financials across multi site portfolios. Delivered in a secure, 21 CFR Part 11 supporting cloud, it connects to Advarra’s eReg/eSource and integrates via API with EHR/other systems.
- Why it stands out
- End to end site financials from budgeting to receivables.
- Enrollment funnel and milestone tracking to accelerate timelines.
- Two way texting and payments for participant engagement.
- Native connectivity to the broader Advarra ecosystem.
- SOC 2 certified infrastructure with audit ready logging.
- Ideal trial fit
Independent sites and health systems running multi study Phase II to III portfolios, particularly where hybrid workflows demand strong recruitment tracking and financial governance. - Pricing snapshot
Quote based cloud subscription with optional modules.
3. EDGE, a CTMS for hospitals and research networks
EDGE standardizes study operations across sites with participant tracking, document control, finance, and reporting. Hosted on Microsoft Azure with security and audit logging, it supports organizational workflows; API/EHR integrations are available subject to vendor validation.
- Why it stands out
- Recruitment dashboards to manage portfolio enrollment.
- Configurable workflows mapped to institutional processes.
- Version controlled document management for governance.
- Finance module for budgets and cost recovery.
- Delegation logs and training tracking to maintain compliance.
- Ideal trial fit
Health systems and research networks coordinating multi site interventional or observational studies that require consistent processes and financial transparency. - Pricing snapshot
Quote based institutional SaaS; request a demo for details.
eRegulatory, eISF/eTMF, and quality management
Maintaining regulatory compliance and inspection readiness is a non negotiable aspect of clinical research, requiring meticulous management of essential documents. This category features software designed to digitize and streamline regulatory workflows, including the electronic Investigator Site File (eISF) and electronic Trial Master File (eTMF). These tools are vital for ensuring quality control, adherence to regulatory standards, and accelerating the study startup process by digitizing essential document workflows.
1. Florence eBinders, a site controlled eISF with remote monitoring
Florence eBinders gives sites a Part 11 compliant eISF/eReg with optional eSource and eConsent, enabling sponsor/CRO remote monitoring via SiteLink. This SaaS platform integrates through open APIs and SSO, connecting to CTMS, EMR/EHR, and sponsor eTMFs to reduce duplication.
- Why it stands out
- Site controlled eISF with compliant eSignatures and audit trails.
- Remote monitoring and SDV to accelerate sponsor oversight.
- Certified copy eSource ingestion from EMR/EHR systems.
- SiteLink API for direct sponsor eTMF connectivity.
- CTMS integrations (e.g., OnCore) to streamline operations.
- Ideal trial fit
AMCs, site networks, and sponsors in decentralized/hybrid studies where certified copy eSource and remote review shorten startup and visits. - Pricing snapshot
Quote based; often sponsor funded for sites.
2. Dot Compliance, a Salesforce native eQMS with CTMS/TMF
Dot Compliance unifies quality and clinical operations on Salesforce with eQMS plus CTMS and eTMF (DIA Reference Model), document control, training, and CAPA. The cloud platform supports 21 CFR Part 11/GxP, leverages native SSO, and integrates broadly via Salesforce APIs.
- Why it stands out
- eTMF aligned to the DIA TMF Reference Model for inspection readiness.
- Combined CTMS/QMS workflows to connect CAPA and trial risks.
- Real time dashboards for portfolio performance tracking.
- AI assistance, pre validation, and native Salesforce integration.
- Ideal trial fit
Sponsors and CROs seeking a QMS first backbone that connects regulatory, quality, and TMF content to maintain global inspection readiness across programs. - Pricing snapshot
Quote based SaaS with packaged offerings; free trial available.
3. Research Manager, an eTMF/eISF and RIMS for audit ready studies
ResearchManager centralizes study documentation for sponsors, CROs, and sites with eTMF, eISF, and RIMS to streamline ethics submissions. The 21 CFR Part 11/GCP compliant, cloud based suite integrates across its modules (EDC, CTMS) via APIs and supports the DIA TMF Reference Model.
- Why it stands out
- Configurable eTMF built on the DIA TMF Reference Model.
- eISF sync keeps site files aligned with sponsor TMF.
- Validated eSignatures with reminders and complete audit trails.
- RIMS to manage submissions and approvals end to end.
- API first architecture to connect the suite and external tools.
- Ideal trial fit
Academic centers, CROs, and sponsors running multi center studies needing strong inspection readiness and cross border document governance. - Pricing snapshot
On Demand and Enterprise options; free trial available.
Recruitment, participant management, and study design
A successful clinical trial begins long before the first patient is enrolled, with robust study design and an effective patient recruitment strategy. The tools in this final section are dedicated to these foundational elements, helping research teams optimize their trial protocols and manage the patient journey. This category includes everything from sophisticated patient registry software to powerful recruitment platforms.
Alongside recruitment, robust study design is paramount. This is where statistical analysis software becomes indispensable. Tools like SAS, R, and the listed nQuery are used by biostatisticians to determine the appropriate sample size, define endpoints, and plan for interim analyses, including complex adaptive trial designs. This statistical foundation ensures the study is scientifically sound and capable of producing valid, interpretable results.
1. Ripple, a recruitment CRM and participant management platform
Ripple helps sites and CROs accelerate enrollment with a recruitment CRM, participant registry, eConsent, and scheduling. Built for HIPAA and 21 CFR Part 11 readiness, it integrates via API/SSO to align with existing CTMS/EDC stacks and orchestrate outreach and follow up.
- Why it stands out
- Recruitment CRM with dashboards to triage and track funnels.
- Cross study registry to build re contactable cohorts.
- Built in eConsent with compliant e signatures and delivery.
- Bulk email/SMS for timely participant communication.
- Calendar sync and tasking for day to day site coordination.
- Ideal trial fit
Sites and CROs in decentralized or hybrid, longitudinal, device, or behavioral health studies that depend on sustained recruitment and retention. - Pricing snapshot
Quote based; available upon request after a sales demo.
2. Studypages, a recruitment hub with eConsent and secure messaging
Studypages gives sponsors, CROs, and sites a modular platform to launch recruitment at scale with study galleries, CRM, secure messaging, eConsent, and optional EDC/ePRO. The HIPAA/GDPR compliant SaaS supports SSO, APIs, and EHR connectivity with 21 CFR Part 11 ready capabilities.
- Why it stands out
- Multilingual prescreeners and branded study pages to widen reach.
- Workspace CRM with automated scheduling and reminders.
- Encrypted, PHI ready chat and study specific VoIP.
- Configurable eConsent with audit logs and version control.
- EDC/ePRO builder with offline mobile capture.
- Ideal trial fit
Multi site academic centers and sponsors running decentralized/hybrid studies that demand unified recruitment, engagement, and site enablement. - Pricing snapshot
Enterprise access is quote based; select modules may offer self serve tiers.
3. nQuery, a sample size and adaptive design powerhouse
nQuery is a specialized trial design tool for sponsor and CRO biostatisticians to calculate sample size, model adaptive designs, and generate randomization lists. Delivered as a validated Windows application, it outputs protocol ready reports and exports for IRT/EDC; it’s not a 21 CFR Part 11 platform.
- Why it stands out
- 1000+ validated procedures for sample size and power.
- Adaptive design simulation: group sequential, MAMS, re estimation.
- Bayesian assurance modules to incorporate prior evidence.
- Enrollment and event forecasting for milestone planning.
- Randomization list generation for downstream IRT/EDC.
- Ideal trial fit
Biostatistics teams designing Phase I to IV drug, device, or diagnostic studies, especially adaptive trials, aiming to right size recruitment and de risk timelines. - Pricing snapshot
Tiered annual named user subscriptions; 14 day free trial.
What’s Next: The Future of Clinical Trials and Software
The future of clinical research software is intelligent, decentralized, and patient centric. We are moving away from siloed systems toward AI native platforms that can predict recruitment bottlenecks, identify data anomalies, and personalize the participant journey.
Decentralized Clinical Trials (DCTs) are becoming the new standard, enabled by software that supports remote monitoring, at home data collection, and virtual site management. This approach not only makes research more accessible for patients but also allows for the collection of more real world data, providing a clearer picture of a treatment’s effectiveness. Companies focused on unified software and service models are leading this charge, simplifying execution for even the most complex protocols.
Conclusion: How to Move Forward
Selecting the right clinical research software is one of the most critical decisions a sponsor or CRO will make. The right platform can significantly accelerate study timelines, improve data integrity, and enhance the experience for both sites and participants. By prioritizing integrated systems that are compliant, scalable, and user friendly, research organizations can overcome operational hurdles and focus on advancing science. As technology continues to evolve, embracing modern, flexible solutions will be key to success in the next generation of clinical development.
To learn how an all in one platform with integrated services can power your next study, explore the solutions offered by Curebase.
FAQ
What is clinical research software used for?
Clinical research software is used to manage and execute all aspects of a clinical trial. This includes study design, patient recruitment, data collection (EDC), patient reported outcomes (ePRO), regulatory compliance, and overall trial management (CTMS).
Is clinical research software regulated?
Yes, software used in clinical trials that collects, manages, or analyzes patient data is highly regulated. In the United States, it must comply with the FDA’s 21 CFR Part 11 requirements for electronic records and signatures.
What is the difference between an EDC and a CTMS?
An Electronic Data Capture (EDC) system is focused specifically on collecting clinical trial data electronically. A Clinical Trial Management System (CTMS) is used for operational management, tracking study milestones, site performance, financials, and regulatory documents. Modern platforms often integrate both functions.
How does software support decentralized clinical trials (DCTs)?
Software is the core enabler of DCTs. It provides the tools for remote participant engagement, including eConsent, telehealth visits, and ePRO data collection via mobile apps. It also helps coordinate at home services like mobile nursing or blood draws.
What should I look for when choosing a clinical research software vendor?
Look for a vendor with a proven track record, strong regulatory compliance, a user friendly interface for all stakeholders, and a flexible platform that can adapt to your specific study design. It is also beneficial to find a partner that offers both software and operational support.
How much does clinical research software cost?
Pricing varies widely based on the complexity of the study, the number of participants, and the specific modules required. Most vendors use an enterprise model with per study licensing fees plus costs for professional services like setup and training.
